 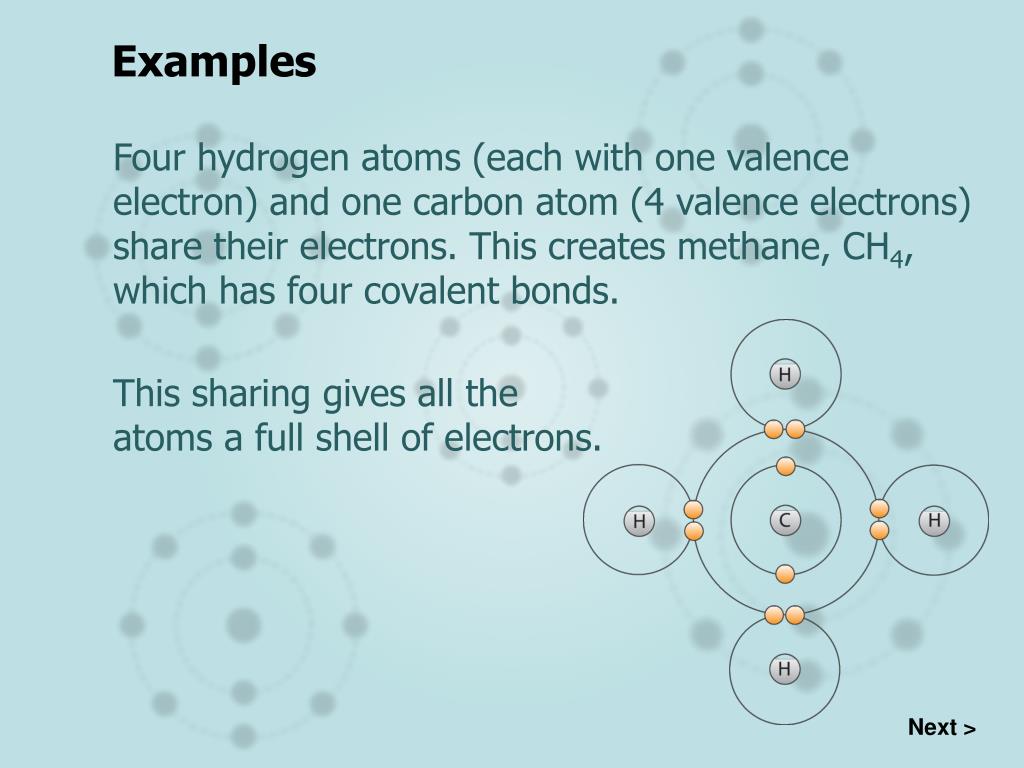
Next, compare the electron groups surrounding the central atom to identify the molecular geometry of BrF5. The bromine atom has one non-bonding lone pair of electrons. Each fluorine atom has three non-bonding lone pairs of electrons. 
PICTURED: Five fluorine atoms are single bonded to one central bromine atom. Thus, a BrF5 molecule has a total of 42 valence electrons, 7+7(5)=42, as shown in the Lewis structure of BrF5. Fluorine and bromine atoms each have seven valence electrons. Begin by counting the number of valence electrons in BrF5.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
